SOLVED: Potassium hydroxide dissociates in water to produce hydroxide ions. KOH(s) + H2O(l) â†' K+(aq) + OH-(aq) Given the information above, how is potassium hydroxide categorized? A. Both a Bronsted-Lowry base and

SOLVED: Potassium hydroxide dissociates in water to produce hydroxide ions. KOH(s) + H2O(l) â†' K+(aq) + OH-(aq) Given the information above, how is potassium hydroxide categorized? A. Both a Bronsted-Lowry base and
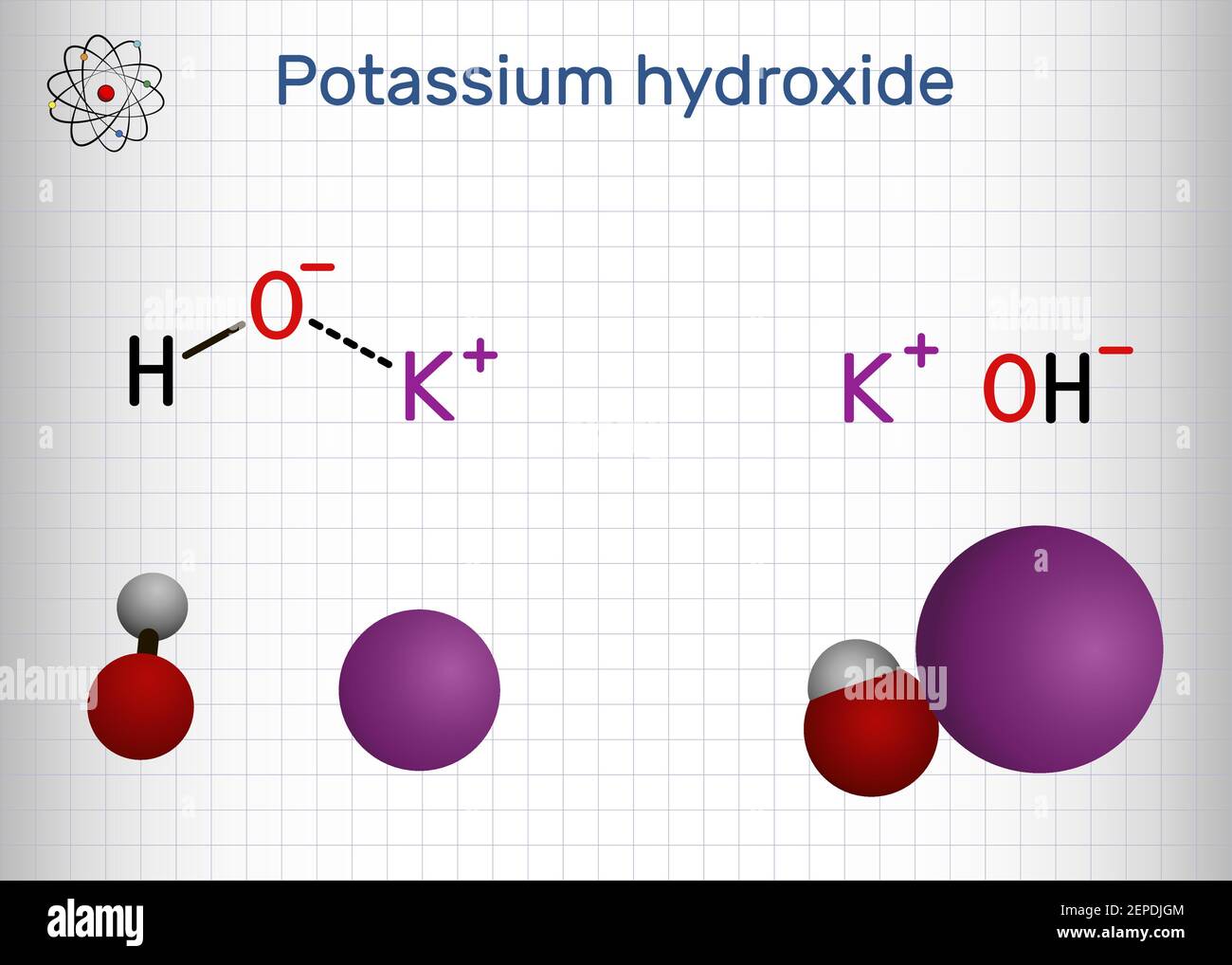
Potassium hydroxide, caustic potash, lye molecule. KOH is strong caustic base and alkali, ionic compound. Structural chemical formula and molecule mod Stock Vector Image & Art - Alamy
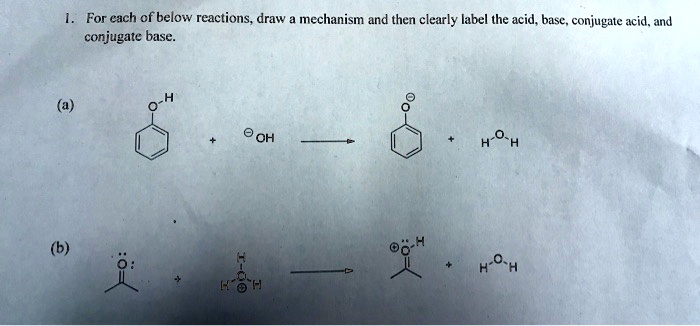
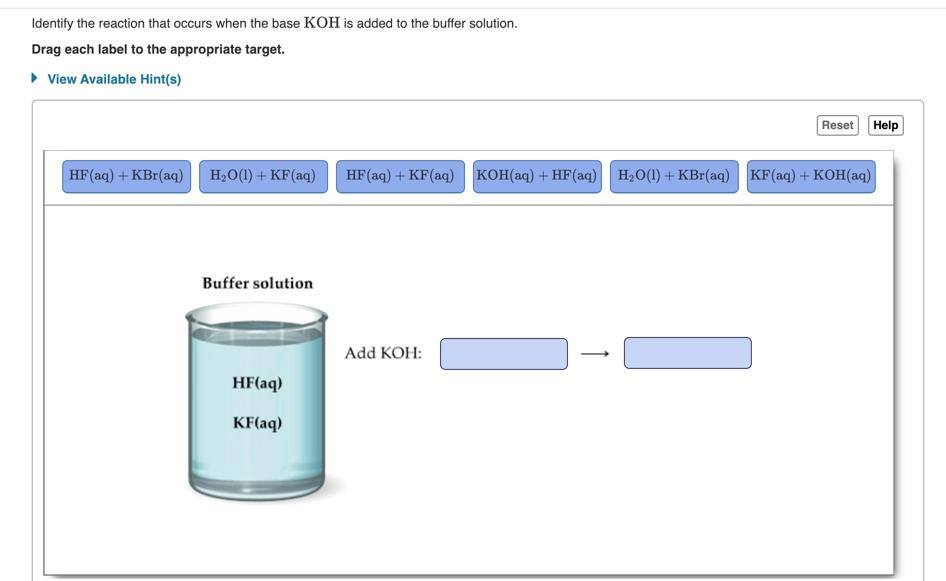
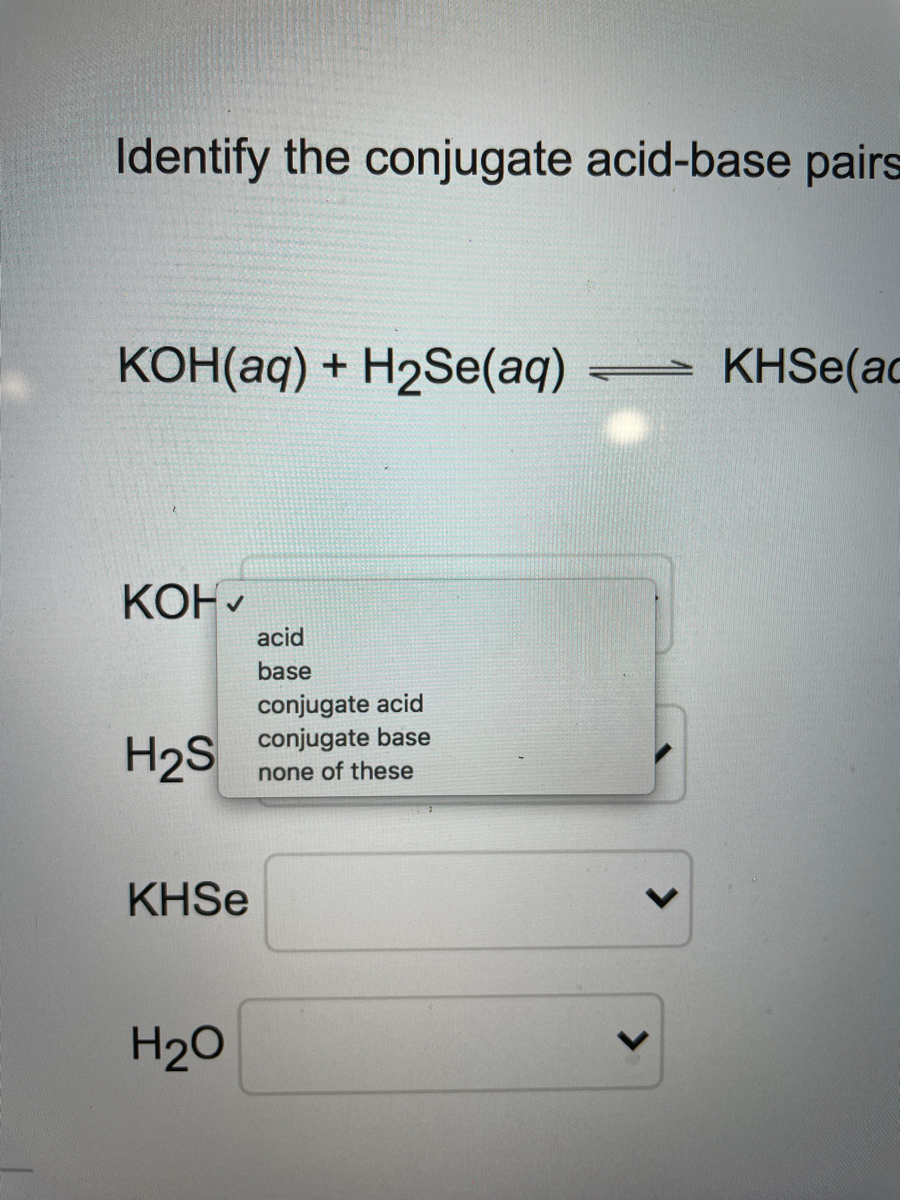
SOLVED: For each of below reactions, draw mechanism and then clearly label the acid; base, conjugate acid, and conjugate base. OH Koh

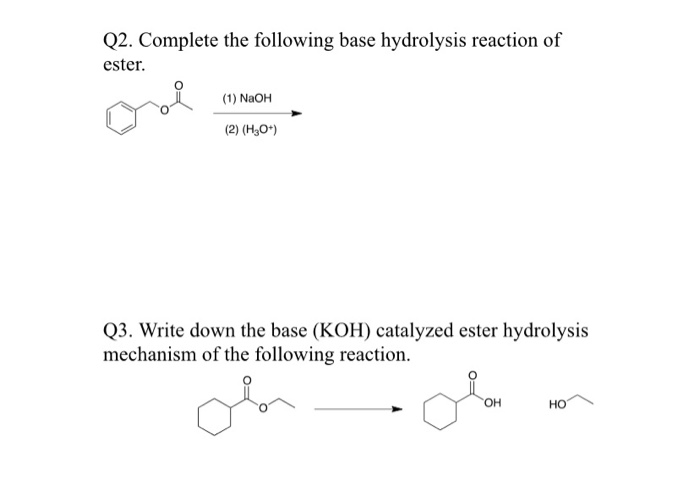
Give the equation for the base hydrolysis of methyl benzoate by KOH. What are the two types of molecules being formed in this reaction? | Homework.Study.com